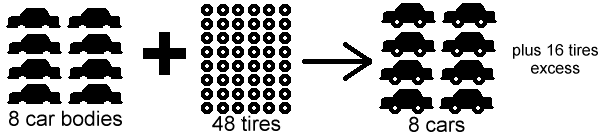Introduction
Limiting reactant is a chemical reaction that limits the amount of product that can be formed. The reaction will stop when all the limiting reactant is consumed.
Ex:
Ex:
Explanation: because there are less car bodies than there are tires and there are four tires on each car only 32 tires are needed. So the car bodies would be the limiting reactant and the tires would be the excess reactant.
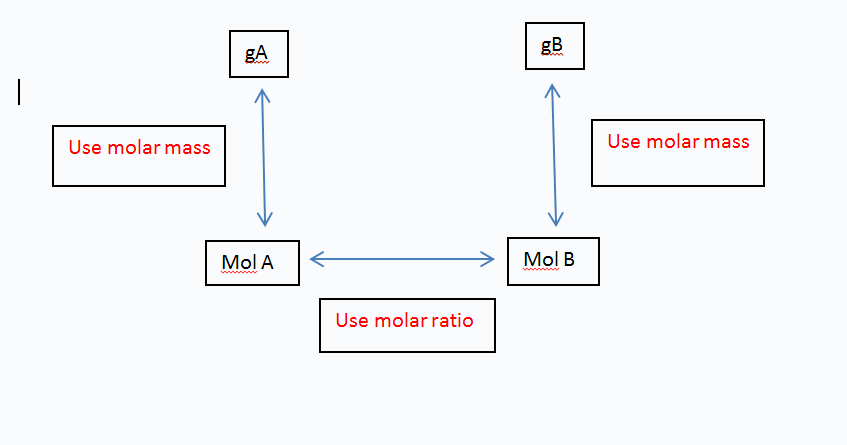
Always remember the mol box!
Lets try it!
Chemistry example:
2Na(s) + 2H2O(l) = 2NaOH(aq) + H2(g)
If there are 10g of each reactant, what's the maximum amount of H2(g) that can be produced?
10g of 2Na x 1mol Na / 23g of Na x 1 mol H2 / 2 mol Na x 2g of H2 / 1 mol H2 = 0.43g of H2
10g of 2H2O x 1 mol H2O / 18g H2O x 1 mol H2 / 2 mol H2O x 2g H2 / 1 mol H2 = 0.56g of H2
When the 10g of 2Na react, 0.43g of H2 are produced
When 10g of 2H2O react, 0.56g of H2 are produced
Therefore 10g of Na is your limiting reactant because it produces a smaller amount of H2 then the 10g of 2H2O
2Na(s) + 2H2O(l) = 2NaOH(aq) + H2(g)
If there are 10g of each reactant, what's the maximum amount of H2(g) that can be produced?
10g of 2Na x 1mol Na / 23g of Na x 1 mol H2 / 2 mol Na x 2g of H2 / 1 mol H2 = 0.43g of H2
10g of 2H2O x 1 mol H2O / 18g H2O x 1 mol H2 / 2 mol H2O x 2g H2 / 1 mol H2 = 0.56g of H2
When the 10g of 2Na react, 0.43g of H2 are produced
When 10g of 2H2O react, 0.56g of H2 are produced
Therefore 10g of Na is your limiting reactant because it produces a smaller amount of H2 then the 10g of 2H2O
Methods for finding the limiting reagent
1, Allow you to find how much of one reactant is needed to react all of the other reactants
Example: C3H8(g) + 5O2(g) = 3CO2(g) + 4H2O(l)
Whats the limiting reagent?
(you have 14.8g of C3H8 and 3.44g of 5O2)
14.8g C3H8 x 1 mol C3H8/44g C3H8 x 5 mol O2/I mol C3H8 x 32g O2/1 mol O2 = 53.8g of O2
but there was only 3.44 grams of O2 to start with, therefore O2 is the limiting reagent.
How many moles of CO2 are produced?
*always use limiting reagent*
3.44g O2 x 1 mol O2/32g O2 x 3 mole CO2/ 5 mol CO2 = 0.0605 moles of CO2
How many grams of water (H2O) are produced?
3.44g O2 x 1 miol O2/32g O2 x 4 mol H2O/5 mol O2 x 18g H2O/1 mol H2O = 1.55 grams of H2O
2. "the really short way"
step 1. grams of reactant 1 to moles of reactant 1
step 2. grams of reactant 2 to moles of reactant 2
step 3. determine mole ratio of moles of R1 over moles of R2
step 4. compare to real mole ratio from balanced equation
EX: step 1. 14.8 g C3H8 x 1 mol C3H8/44g C3H8 = 0.336 moles of C3H8
step 2. 3.44 g O2 x 1 mol O2/32g O2 = 0.108 moles of O2
step 3. 0.336 moles C3H8/0.108 moles O2 = 3.11 moles C3H8/ 1 mol O2
step 4. 3.11 moles C3H8/ 1 mol O2 Vs. 1 mol C3H8/5 mol O2
Conclusion: for every 3.11 moles of C3H8 you will need 15 moles of O2, but you don't have that when we look at the original equation. So O2 is the limiting reagent.
Example: C3H8(g) + 5O2(g) = 3CO2(g) + 4H2O(l)
Whats the limiting reagent?
(you have 14.8g of C3H8 and 3.44g of 5O2)
14.8g C3H8 x 1 mol C3H8/44g C3H8 x 5 mol O2/I mol C3H8 x 32g O2/1 mol O2 = 53.8g of O2
but there was only 3.44 grams of O2 to start with, therefore O2 is the limiting reagent.
How many moles of CO2 are produced?
*always use limiting reagent*
3.44g O2 x 1 mol O2/32g O2 x 3 mole CO2/ 5 mol CO2 = 0.0605 moles of CO2
How many grams of water (H2O) are produced?
3.44g O2 x 1 miol O2/32g O2 x 4 mol H2O/5 mol O2 x 18g H2O/1 mol H2O = 1.55 grams of H2O
2. "the really short way"
step 1. grams of reactant 1 to moles of reactant 1
step 2. grams of reactant 2 to moles of reactant 2
step 3. determine mole ratio of moles of R1 over moles of R2
step 4. compare to real mole ratio from balanced equation
EX: step 1. 14.8 g C3H8 x 1 mol C3H8/44g C3H8 = 0.336 moles of C3H8
step 2. 3.44 g O2 x 1 mol O2/32g O2 = 0.108 moles of O2
step 3. 0.336 moles C3H8/0.108 moles O2 = 3.11 moles C3H8/ 1 mol O2
step 4. 3.11 moles C3H8/ 1 mol O2 Vs. 1 mol C3H8/5 mol O2
Conclusion: for every 3.11 moles of C3H8 you will need 15 moles of O2, but you don't have that when we look at the original equation. So O2 is the limiting reagent.
| limiting_reagents.pdf | |
| File Size: | 64 kb |
| File Type: | |
If you would like more practice, click "Extra Practice"!