Stoichiometry is the word we use to describe the calculations used to determine products and reactants in a reaction.
To begin, we must first have a chemical equation.
Let's use:
AgNO3 (aq) + NaCl (aq) --> AgCl (aq) + NaNO3 (aq) or Silver nitrate reacting with Sodium cholride to form Silver chloride and Sodium nitrate (all are aqueous).
Before we start anything we need to make sure that the equation is balanced, which it is here.
Looking at the reaction above we must first establish the mole ratios.
Since they're are no co-efficients in this equation, every reactant has a ratio of 1:1 with its product.
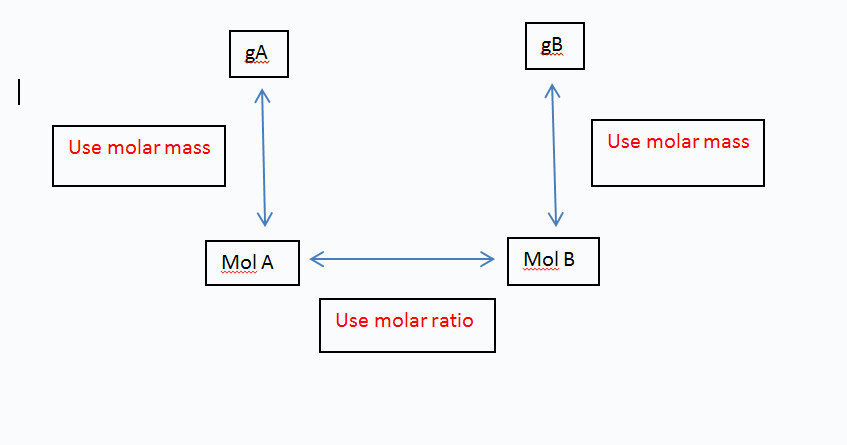
Then we use the mole box to determine what we have and what we are being asked to find.
To begin, we must first have a chemical equation.
Let's use:
AgNO3 (aq) + NaCl (aq) --> AgCl (aq) + NaNO3 (aq) or Silver nitrate reacting with Sodium cholride to form Silver chloride and Sodium nitrate (all are aqueous).
Before we start anything we need to make sure that the equation is balanced, which it is here.
Looking at the reaction above we must first establish the mole ratios.
Since they're are no co-efficients in this equation, every reactant has a ratio of 1:1 with its product.
Then we use the mole box to determine what we have and what we are being asked to find.
To use our reaction above as an example, let's say we start with 3 moles of AgNO3, how many moles of AgCl is being produced?
3 mol AgNO3 x 1 mol AgCl
___________ = 3 mol AgCl
1 mol AgNO3
To find this we simply found what we had (mol A) and what we needed to find (mol B). We then just traced the path needed to find our product's number of moles.
Here's some extra practice.
3 mol AgNO3 x 1 mol AgCl
___________ = 3 mol AgCl
1 mol AgNO3
To find this we simply found what we had (mol A) and what we needed to find (mol B). We then just traced the path needed to find our product's number of moles.
Here's some extra practice.
| stoichiometry-worksheetanswers.pdf | |
| File Size: | 82 kb |
| File Type: | |