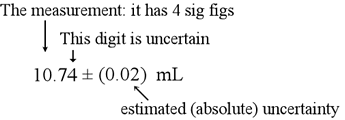Significant Figures and Uncertainty
SIGNIFICANT FIGURES:
Significant figures are the digits of a number that carry precision. We only use sig figs with measurements, NOT WITH CONSTANTS.
Ex: a=(bxh)/2 does not need sig figs but length of table= 2.63 m does!
Rules for sig figs:
1) All numbers from 1 through 9 are significant
Ex: =45.788, 5 sig figs =39.23, 4 sig figs
2) Zeros between 2 sig figs are significant.
Ex: =5.609, 3 sig figs
3) Zeros that are placeholders are NOT significant.
Ex: =0.0052, 2 sig figs because 5.2x10^-3 =260000000, 2 sig figs because 2.6x10^8
4) Zeros appearing after a decimal point are significant. DECIMALS ARE CONSIDERED S.F.
Ex: =10.300, 5 sig figs =77.07777, 7 sig figs
Addition and Subtraction of Sig Figs
When adding and subtracting, we take the SMALLEST amount of decimals and that is the number of decimal places in our answer. We proceed using normal addition and subtraction for the first part.
Ex: 6.7+4.509888= 11.2
Ex: 6.7+4.509888= 11.2
Multiplication and Division of Sig Figs
For the multiplication or division of sig figs, we calculate the amount of significant figures in each number and we use the smallest amount of sig figs for our answer.
| worksheet_sig_fig_9_11_08.pdf | |
| File Size: | 153 kb |
| File Type: | |
Uncertainty
There are 2 types of uncertainty, absolute and relative error.
Absolute: Used mainly in experimentation when you have measured values. You take the measurement you received at the end and then smallest graduation of whatever you were measuring. Then you divide the smallest graduation by your measurement and you have your absolute value. It should end up looking like this:
Absolute: Used mainly in experimentation when you have measured values. You take the measurement you received at the end and then smallest graduation of whatever you were measuring. Then you divide the smallest graduation by your measurement and you have your absolute value. It should end up looking like this:
Next we have relative uncertainty. First we must find our absolute uncertainty. Then we take the absolute uncertainty and divide by our measured quantity and multiply our answer by 100 to obtain at percentage.
Extra practice?
Extra practice?
| ph191_uncert_work.pdf | |
| File Size: | 82 kb |
| File Type: | |